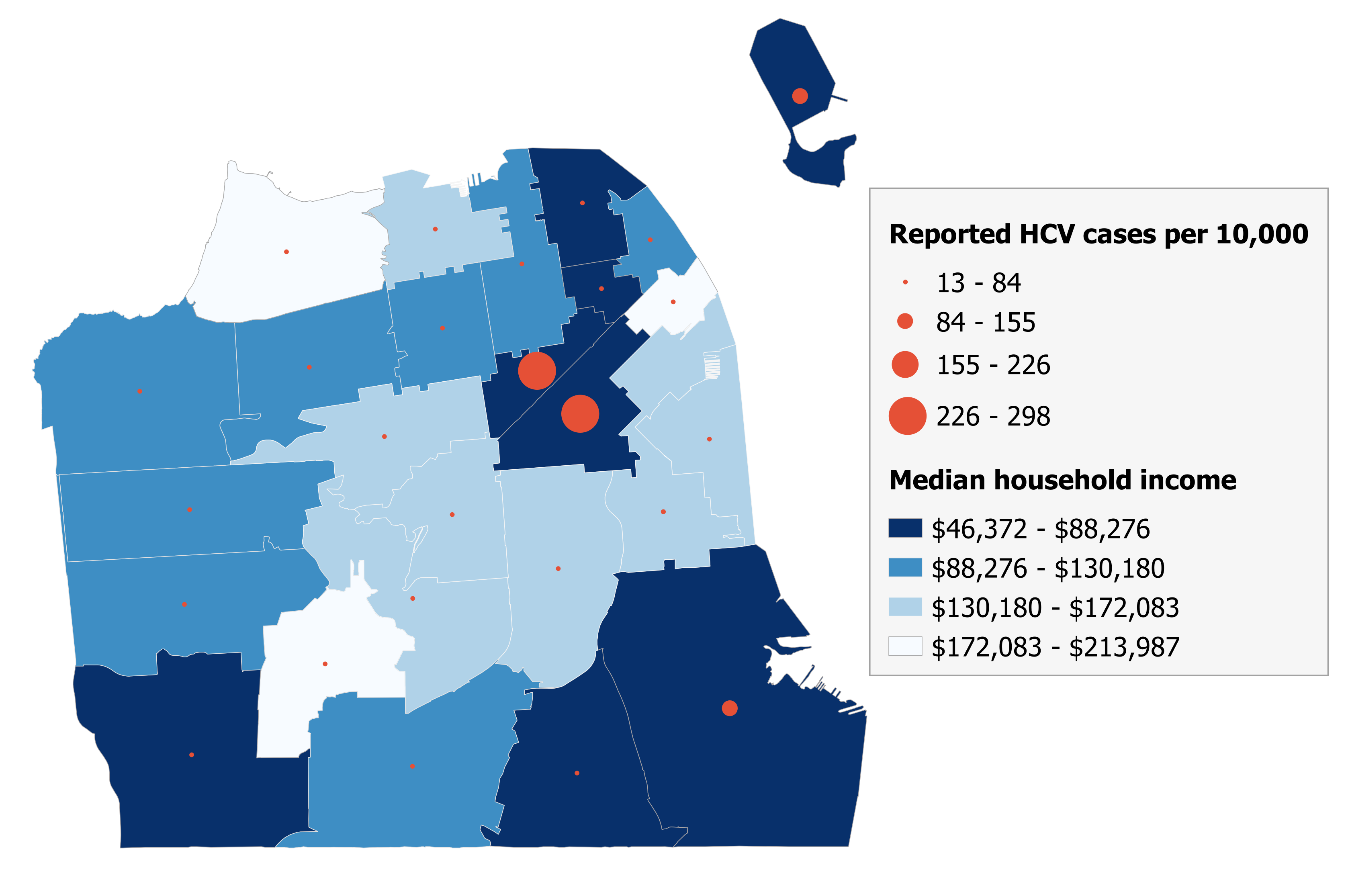
Facente Consulting was hired during the formation of End Hep C SF to provide “backbone support” for this collective impact initiative, assist with strategic planning, and lead research and surveillance activities to monitor progress toward hepatitis C elimination.
Project Descriptions
Collective Impact Backbone Support and Data Analysis to Eliminate Hepatitis C
Harm Reduction Readiness Assessment in Southeast San Diego
County of San Diego Public Health Services
Impact Evaluation of a Social Connection Intervention to Improve LGBTQ+ Mental Health
San Francisco Community Health Center
Evaluating Naloxone Boxes to Prevent Overdoses in Michigan
Harm Reduction Michigan
Test and Be Treated (TBT) Study Data Analysis
University of California, San Francisco (UCSF)
Support for Viral Hepatitis Surveillance Planning, Data Analysis, and Reporting
San Francisco Department of Public Health ARCHES Branch
Planning and Facilitation of Institutes to Improve HIV PrEP Capacity in Florida
SFDPH Center for Learning and Innovation
Tobacco Free Project Quantitative Data Analysis
San Francisco Tobacco-Free Project
Project Coordination and Analytic Support to Improve Global HIV Incidence Estimation
The Consortium for the Evaluation and Performance of HIV Incidence Assays (CEPHIA)
Research Coordination and Data Analysis for an NIH R21 Study on Hepatitis C Epidemiology
University of California, San Francisco (UCSF) - CFAR/Gladstone Institute
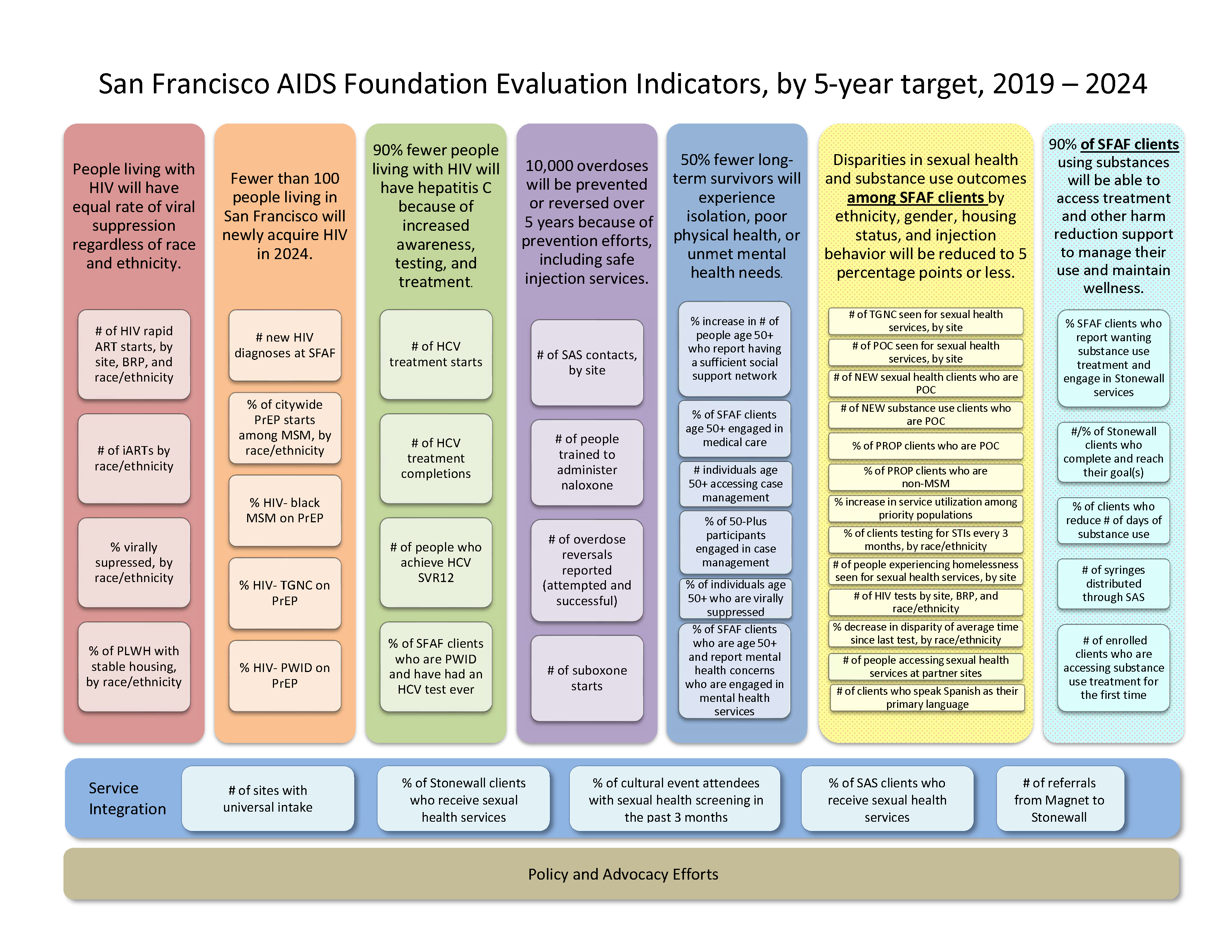
Development and Implementation of Organizational Impact Evaluation
San Francisco AIDS Foundation
Evaluating and Communicating the Impact of Syringe Service Programs Providing Access to MAT
The Center at Sierra Health Foundation
Evaluation for CDC-Funded Workforce Development for San Francisco Department of Public Health
SFDPH Center for Learning and Innovation
Formal Assessment of the Landscape of Medications for Addiction Treatment (MAT) in California
The Center at Sierra Health Foundation
Assistance to Develop a System of Panel Management within a Jail Medical System
SFDPH Jail Health Services
National HIV Behavioral Surveillance (NHBS) Data Analysis
CDPH Office of AIDS
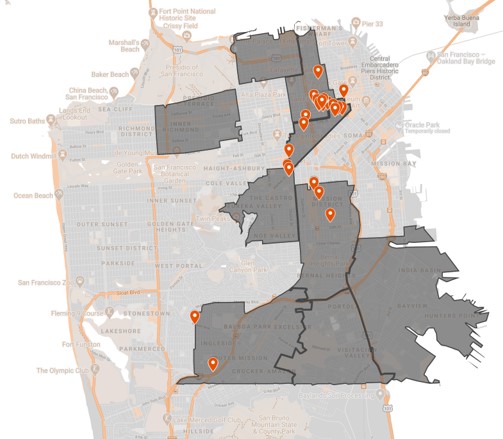
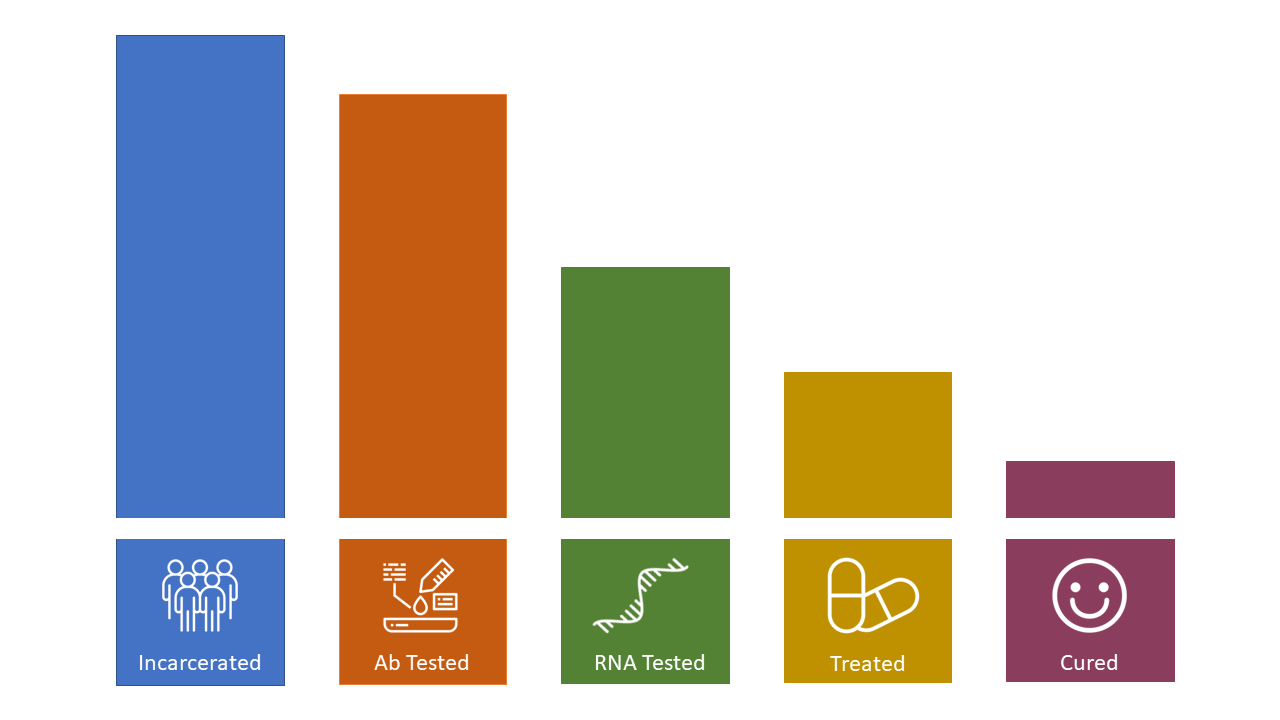
Needs Assessment of Hepatitis C Testing and Treatment in the Jail System
End Hep C SF
Facente Consulting was selected by End Hep C SF to design and lead a mixed-methods needs assessment to identify gaps in hepatitis C (HCV) testing and treatment in San Francisco jails, identify best practices for treatment and linkage, and develop a HCV care cascade for people currently or recently incarcerated.
Statistical Analysis to Support Evaluation of a LGBTQ+ Mental Health Intervention
LGBTQ Connection
Statistical Support for an Advanced HIV Prevention Clinical Trial Design
Gilead Sciences
Facente Consulting supported Gilead Sciences with statistical analysis relating to HIV incidence rates, assisting with the study design, analysis plan, and interpretation of study data for the PURPOSE trials–ultimately leading to the groundbreaking FDA approval of the twice-yearly PrEP medication Lenacapavir.